RESEARCH NEWS
Wollastonite-Enabled Fermentation System Boosts Biohydrogen Yield and Captures CO2
Researchers from the Institute of Applied Ecology of the Chinese Academy of Sciences have developed a fermentation process that simultaneously enhances biohydrogen production and sequesters carbon dioxide within a single system.
The study, led by Dr. LI Weiming, was published in Chemical Engineering Journal.
Dark fermentation produces hydrogen gas from organic substrates under oxygen-free conditions and is regarded as a promising route toward carbon-neutral hydrogen. In practice, however, the process is often limited by the accumulation of volatile fatty acids, which drives down pH and suppresses microbial activity. Conventional pH control relies on alkaline chemicals such as sodium hydroxide, but these agents can cause localized pH spikes, progressive salinity buildup, and require continuous dosing — all without offering any added environmental benefit. A further challenge is that the resulting biogas contains a substantial fraction of CO2, typically requiring energy-intensive downstream separation. Together, these constraints have made it difficult to achieve efficient hydrogen production and carbon mitigation within a single system.
To overcome these limitations, the team introduced wollastonite (CaSiO3), a naturally occurring silicate mineral, as a dual-function additive. As acids accumulate during fermentation, they gradually dissolve the mineral, consuming protons and releasing calcium ions. This mechanism provides continuous, self-regulating pH buffering that stabilized the system at pH 6.5–7.0. At an optimal dosage of 10 g/L, the lag phase of hydrogen production was shortened by roughly 50%, and the specific hydrogen yield increased by approximately 33%.
The stabilized pH environment also reshaped the metabolic landscape. Acetate production rose markedly while lactate accumulation dropped to negligible levels, and the acetate-to-butyrate ratio increased from 0.55 to 0.91 — indicating a shift toward acetate-type fermentation, a pathway stoichiometrically more favorable for hydrogen generation. Microbial community analysis corroborated this shift: the relative abundance of Clostridium sensu stricto 1, a key hydrogen-producing genus, rose from 47.2% to 62.4%, whereas Lactobacillus, associated with competing lactate production, was nearly eliminated.
The study further revealed a trade-off between the two target functions. Efficient CO2 mineralization requires a neutral to slightly alkaline pH, achievable at higher wollastonite dosages (≥15 g/L), yet these conditions compromised hydrogen yield. To decouple the two objectives, the team devised a two-stage strategy: the first stage employs the optimal 10 g/L dosage to maximize hydrogen output, while the second stage applies a post-fermentation pH adjustment to 7.0 to induce carbonation. This approach sequestered 0.49 ± 0.05 L of CO2 per liter of medium and enriched the hydrogen content of the final biogas to 58.2 ± 1.1%. Solid-phase characterization confirmed that the captured CO2 was mineralized as calcite-phase calcium carbonate, a stable form suitable for long-term carbon storage.
A life cycle assessment validated the environmental advantages of the optimized process. Total electricity demand fell from 59.2 to 37.4 MJ per kilogram of hydrogen produced, with lower impacts across all ten evaluated categories, including global warming potential. The findings provide a proof-of-concept for integrating green hydrogen production with in-situ carbon capture in a single biorefinery framework, offering a pragmatic pathway toward negative-carbon biohydrogen.
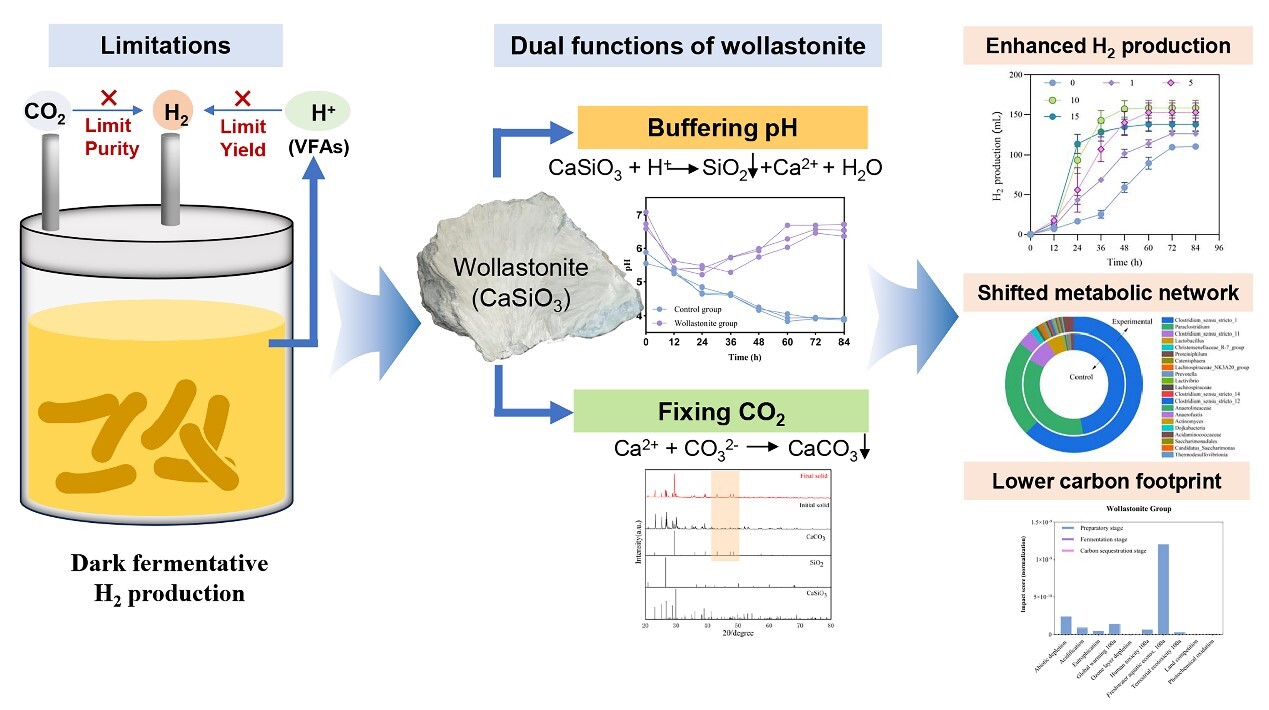
Figure 1. Mechanisms by which wollastonite enhances hydrogen production and enables CO₂ sequestration (Image by LI Weiming).


