Researchers Distinguish the Contribution of Nitrification and Denitrification to Soil NO Production with Its 15N Natural Abundance
Nitric oxide (NO) is readily oxidized in the atmosphere to form nitrate (NO3-), and further cause the atmospheric haze pollution, which severely impacts air quality and human health. In addition, NO is highly reactive and plays an important role in atmospheric chemistry by influencing the production and destruction of ozone and thereby the oxidizing capacity of the atmosphere. To reduce the atmospheric pollution, it is crucial to identify the sources of NO for a given region at a given time. Stable nitrogen (N) isotope technique has been widely used to trace the sources of atmospheric N gases. Soil emission is the second important source of NO, while studies on 15N abundance in NO produced in soil are limited, resulting in large uncertainties of source partition. Nitrification and denitrification are two predominant processes to produce NO in soil, up to now, however, there have been no N isotope fractionation being reported for soil emitted NO from these two processes.
Prof. Fang Yunting and his research group, from the Institute of Applied Ecology, Chinses Academy of Sciences, collected seven soil samples from seven sites including two agricultural lands, two forests and three grasslands.
They detected the 15N natural abundances of NO and substrates (NH4+ and NO3-) under aerobic and anaerobic incubations respectively. Based on the dynamic of 15N abundances and concentrations of products and substrates, they calculated the isotope fractionation factors during nitrification and denitrification.
They found that 15N abundance of NO formed under the aerobic condition (nitrification dominantly) was significantly lower than that under the anaerobic condition (denitrification dominantly) (Fig. 1). The isotope fractionation factors under the aerobic condition were significantly larger than that under the anaerobic condition (Fig. 2), regardless of soil types. From their findings, it is possible to distinguish the relative contribution of nitrification and denitrification to soil NO production by means of 15N natural abundance of NO, thus providing theoretical basis for N cycle model. Additionally, they found that 15N abundance of NO produced in soil is significantly smaller than that from fossil fuel and biomass combustion (Fig. 1). As a result, the contribution of soil emission and anthropogenic sources to atmospheric NOx is likely distinguished with the N stable isotope technique.
This study is supported by the National Key Research and Development Program of China, the Key Research Program of Frontier Sciences of Chinese Academy of Sciences, and Hundred Talents Program of Chinese Academy of Sciences.
This study entitled “δ15N of nitric oxide produced under aerobic or anaerobic conditions from seven soils and their associated N isotope fractionations” has been published in Journal of Geophysical Research-Biogeosciences.
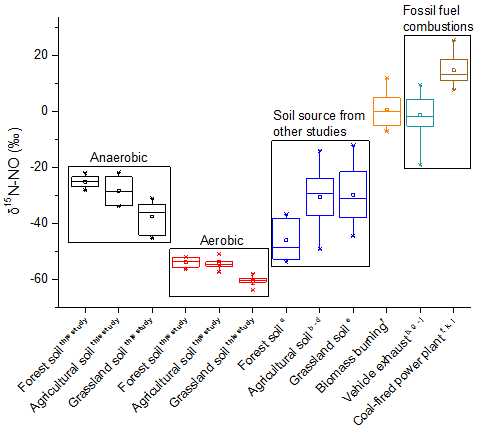
Fig 1. The δ15N values of the NO produced from different sources (Image by KANG Ronghua).
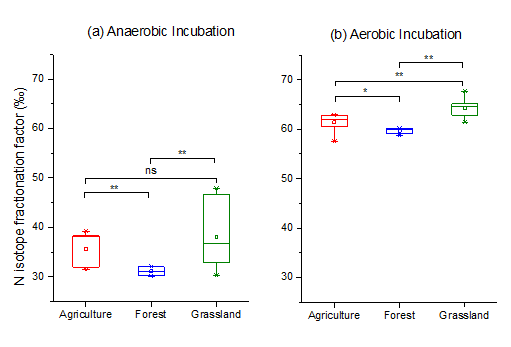
Fig 2. The difference of N isotope fractionation for NO production among (Image by KANG Ronghua).
Publication Name: KANG Ronghua et al.
Email: yueqian@iae.ac.cn



