Study Reports Sustained Pulmonary Injury Induced by Repeated Exposure to Polystyrene Nanoplastics in Mice
Researchers from the Institute of Applied Ecology of the Chinese Academy of Sciences and Shengjing Hospital of China Medical University have examined the respiratory effects of inhaled nanoplastics and found that repeated exposure to nanoplastics can induce sustained pulmonary injury in mice.
The study was published in Ecotoxicology and Environmental Safety.
Nanoplastics are extremely small plastic particles typically less than 1 μm in size. Due to their low density, small particle diameter, and large surface area, they can remain suspended in the atmosphere for extended periods and be easily transported over long distances. These characteristics enable them to be inhaled through the respiratory system and subsequently deposited in lung tissue, posing potential risks to respiratory health. However, the toxic effects and underlying mechanisms of inhaled nanoplastics remain insufficiently understood, particularly under conditions of continuous exposure and the potential for persistent toxicity after exposure cessation.
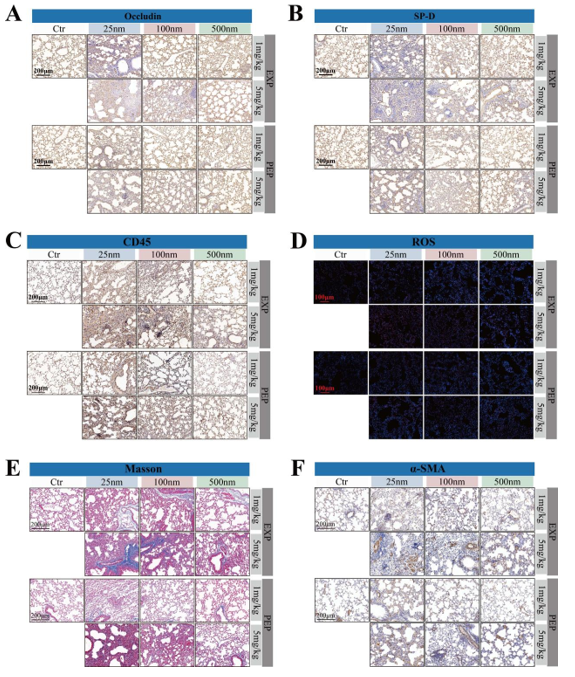
To investigate these questions, the study, led by Dr. XU Mingkai and Prof. LI Tiegang, established a mouse model of polystyrene nanoplastics (PS-NPs) inhalation exposure via intratracheal instillation. C57BL/6 mice (6–8 weeks old) were exposed to PS-NPs with particle sizes of 25, 100, and 500 nm. The experimental design included a 4-week exposure period (EXP) followed by a 2-week post-exposure period (PEP). Exposure doses were set at 1 mg/kg and 5 mg/kg.
The researchers found that inhaled nanoplastics accumulated in the lung tissue of mice, translocated to distant organs, including the heart, liver, spleen, and kidneys, and reduced peripheral white blood cell counts. The study further showed that nanoplastic exposure disrupted the alveolar epithelial barrier, induced inflammatory responses and oxidative stress in lung tissue, impaired lung function, and promoted collagen deposition, thereby promoting the progression of pulmonary fibrosis. Pulmonary fibrosis is a progressive and chronic lung disease characterized by alveolar epithelial barrier injury and excessive collagen deposition, gradually impairing respiratory capacity.
The team also reported that toxic effects were strongly size-dependent, with smaller particles inducing greater toxicity. Moreover, adverse effects were sustained during the PEP, indicating that PS-NPs–induced injury was not readily reversible in the short term.
Further analysis indicated that macrophage polarization may play a role in the progression of PS-NPs–induced pulmonary fibrosis. Macrophages are key effector and regulatory cells in particle-induced pulmonary pathology, exhibiting high plasticity and the ability to polarize into classically activated (M1) and alternatively activated (M2) phenotypes, which are closely associated with the progression of pulmonary fibrosis. The researchers observed a shift in pulmonary macrophages from the M1 to the M2 phenotype, suggesting that this transition may contribute to the development of fibrosis following nanoplastic exposure.